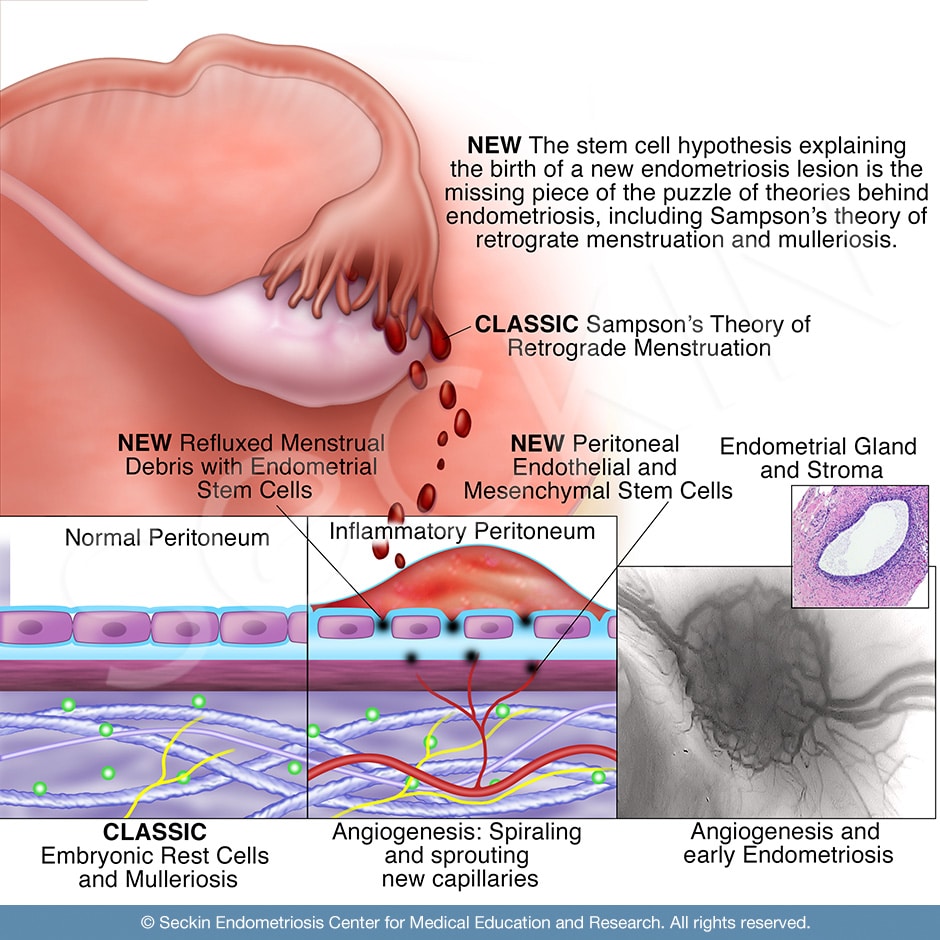
Science can’t point to a singular cause of endometriosis. However, there are a multitude of highly supported theories that exist. These range from the classic Sampson’s theory of retrograde menstruation to more modern theories such as the stem cell theory. It is most likely that the true cause of endometriosis is a combination of these well-researched theories.
What is endometriosis?
Endometriosis is a disease in which endometrial-like tissue forms outside the uterus. Lesions are estrogen-dependent, benign, inflammatory, and stem-cell driven. At times, they are progressive with diffuse fibrosis, deep infiltration, and resistance to apoptosis and progesterone. While the exact cause of endometriosis is unknown, multiple plausible theories have been proposed. These include retrograde menstruation, implantation, spontaneous development of ectopic endometrial tissue (mülleriosis), peritoneal metaplasia, and differentiation of mesenchymal cells.
Get a Second Opinion
Our endometriosis specialists are dedicated to providing patients with expert care. Whether you have been diagnosed or are looking to find a doctor, they are ready to help.Our office is located on 872 Fifth Avenue New York, NY 10065.
You may call us at (212) 988-1444 or have your case reviewed by clicking here.
The stem cell theory
The normal human endometrial lining is subject to numerous cycles of growth and differentiation as the endometrium sheds and regenerates during a normal menstrual cycle. Its regenerative capacity is believed to come from resident mesenchymal stem/stromal cells. These potentially have the classic properties of bone marrow mesenchymal stem cells. They are multipotent (able to give rise to several cell types) and have the potential to self-renew and reconstitute in ectopic locations. According to the stem cell theory, it is this ability that leads to the development of endometriosis.
The role of genetics
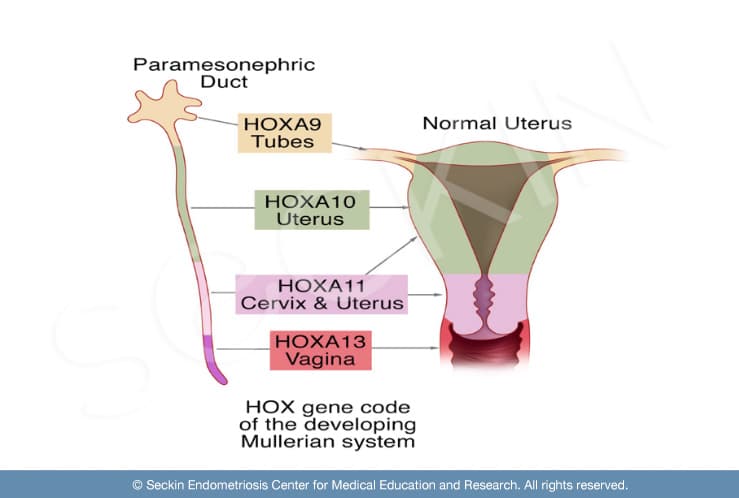
Genealogical databases have shown a familial connection between first and second-degree relatives affected by endometriosis. This suggests a genetic component of the disease. In addition, alterations in HOX gene expression cause uterine structural abnormalities and abnormal endometrial development. Gene expression profiling has shown differences in the endometrium with a higher susceptibility for endometriosis. The molecular composition of the endometrial tissue influences whether ectopic endometrium will have the ability to interact with a receptive peritoneum (lining of the abdominal cavity) and become functional, thereby leading to disease. For example, a low expression of the HOXA10 gene in eutopic endometrium is present in animal models of endometriosis and in women with the disease. These findings suggest that genetics may play a role in endometriosis development.
A genetic predisposition to the disease could account for the discrepancy between those who develop endometriosis lesions or symptoms and those who do not. For instance, many women experience retrograde menstruation (the backward flow of menstrual debris). This is evidenced by blood in the pelvis at entry during laparoscopy. However, most of these women are asymptomatic or have no pathologic evidence of endometriosis.
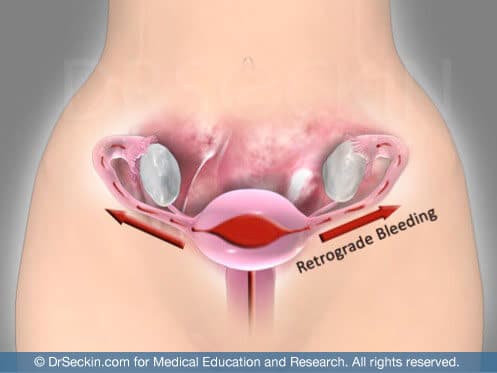
The theory of retrograde menstruation
During normal menstruation, the female body naturally sheds the endometrium. This is the tissue that grows every month to prepare for the implantation of a fertilized egg. While many women experience retrograde menstruation, there is speculation that this mechanism may, in fact, play a role in endometriosis development. Sampson’s theory of retrograde menstruation is based on endometrial cells being transported through the fallopian tubes into the peritoneal cavity at the time of menstruation, leading to the development of endometriosis.
As a woman’s hormones change during her menstrual cycle, these endometriosis implants (also referred to as lesions or nodules) caused by retrograde menstruation, ovarian development, and leakage respond to hormonal fluctuations. The lesions grow, menstruate, and shed along with the normal lining of the uterus, causing endometriosis to spread. Unlike normal endometrium that naturally leaves the body, when these implants shed, there is no way for the material to exit. This is because the lesions implant in ectopic regions much deeper in the pelvic and abdominal cavities.
The potential role of uterine structural anomalies
Multiple studies have also noted an association between uterine anomalies and endometriosis, a relationship that may in fact be causal. Structural defects of the uterus likely contribute to dysfunctional uterine contractions and abnormal blood flow during menstruation, i.e. retrograde menstruation. Both obstructive and nonobstructive uterine anomalies can lead to increased retrograde menstruation. However, the persistence of endometriosis symptoms after the correction of an outflow obstruction challenges the theory that an outflow obstruction must be present to cause abnormal menstruation. Nevertheless, it could contribute to an increased volume of blood directed through the fallopian tubes into the peritoneal cavity. The correction of structural abnormalities can improve endometriosis symptoms, and possibly a common sequela of both uterine anomalies and endometriosis: infertility.
The potential role of abnormal uterine peristalsis
A distinct but interrelated issue to uterine structure abnormalities is dysfunctional uterine peristalsis and the distribution of ectopic endometrial tissue. Uterine peristalsis occurs in the endometrium and subendometrial myometrium. Women with laparoscopically proven endometriosis show enhancement of the subendometrial myometrium on transvaginal ultrasound. Functional MRI studies demonstrate similar findings of dysfunctional peristalsis. This suggests that the origin of endometriosis could be in the structure and function of the uterus. With a structural anomaly, the uterus will be at an increased risk of dysfunctional uterine peristalsis. The combination of the presence of dysperistalsis and retrograde menstruation with a receptive peritoneum to this menstrual debris could explain how ectopic endometrial tissue can anchor and become functional, thus causing endometriosis.
Symptoms of endometriosis
Endometriosis causes internal bleeding, inflammation, and the expression of irritating enzymes. It can implant in the bowel, bladder, intestines, and other areas of the pelvic cavity, causing severe pain, the formation of scar tissue, and adhesions (fibrous bands of dense tissue). It can also cause the growth of new blood vessels (angiogenesis) and root to existing nerves. This can lead to chronic pelvic pain, severe cramping, bowel or urinary disorders, painful intercourse, and even infertility.
If you find yourself experiencing these symptoms, it could be due to a high rate of retrograde menstruation. This could mean that endometriosis may be either forming or has already formed. The most important step towards treating your symptoms is through overcoming the taboo that surrounds the menstrual cycle and in turn endometriosis itself.
Get a Second Opinion
Our endometriosis specialists are dedicated to providing patients with expert care. Whether you have been diagnosed or are looking to find a doctor, they are ready to help.Our office is located on 872 Fifth Avenue New York, NY 10065.
You may call us at (646) 960-3080 or have your case reviewed by clicking here.
Dr. Seckin is an endometriosis specialist and women’s reproductive health advocate. He has been in private practice for over 30 years at Lenox Hill Hospital with a team of highly skilled personnel.
Dr. Seckin specializes in advanced laparoscopic procedures and is recognized for his expertise in complex cases of deep infiltrating endometriosis of the pelvis. He is particularly dedicated to performing fertility-preserving surgeries on cases involving the ovaries.
He has developed patented surgical techniques, most notably the “Aqua Blue Excision” technique for a better visualization of endometriosis lesions. His surgical techniques are based on precision and microsurgery, emphasizing organ and fertility preservation, and adhesion and pain prevention.
Dr. Seckin is considered a pioneer and advocate in the field of endometriosis.
