Definitive Surgery for Endometriosis after Completion of Family Planning
By: Tamer A. Seckin | Serin Seckin | Harry Reich
4.8.1 Introduction and Background
Endometriosis is a condition characterized by the presence of endometrial glands and stromal cells surrounded by reactive inflammatory fibrosis outside the inner layer of the uterus. This definition includes both adenomyosis, which is the infiltration of endometrial cells deeper into the myometrium, and inflammatory fibrosis of peritoneal layers outside the uterus.15 A clear clinical understanding of the disease is necessary for definitive surgical treatment. Treatment can be achieved by complete excision of endometriotic fibrosis and implants.
It is estimated that endometriosis affects 6–10 % of reproductive-aged women and causes cyclic pain, infertility, and loss of quality of life. Often these patients undergo multiple repetitive surgeries. According to a prospective 4-year longitudinal study in North America, 27 % of patients were reoperated on after initial endometriosis surgery, and 12 % of all patients initially operated on (44 % with subsequent surgery) ultimately received a hysterectomy.28
Inflammatory endometriotic fibrosis, which is estrogendependent, can cause mild to severe changes in the morphology of the peritoneal membrane and the topography of the cul-de-sac and pelvic sidewalls. Superficial fibrosis and dense adhesions restrict the functions of pelvic reproductive and non-reproductive organs. This causes the characteristic, healthy texture of the peritoneum – which is normally shiny, slippery, and transparent – to change. Deep fibrotic involvement of the retroperitoneum presents an additional challenge in both visual recognition and surgical treatment. Menorrhagia with cyclic and chronic pelvic pain is the most recognizable symptom of endometriosis, along with dyschesia, dyspareunia, and constipation.
For women with endometriosis who no longer wish to have children, the definitive option for recurrent symptoms after conservative surgery is total hysterectomy with bilateral oophorectomy and excision of visually recognized and suspected endometriosis tissues. Depending on the excision expertise of the surgeon, ovarian conservation may be considered.18 According to the American College of Obstetricians and Gynecologists, ‘… surgeons should consider removing both fallopian tubes and spare the ovaries in women at low risk for ovarian cancer.’2
Endometriosis primarily affects the pelvic peritoneum in the first order and most of the signs and symptoms that make the disease elusive are secondary to peritoneal involvement. However, it is often said that endometriosis is a disease of the uterus and the most commonly accepted etiology is believed to be retrograde menstrual debris refluxed from the fallopian tubes.6 According to the works of Taylor,11 and Gargett,14 stem cells of the endomyometrial junction and their further stimulation of angiogenesis and pelvic peritoneum are possibly the modern explanation that complements and brings together older theories of mulleriosis and retrograde menstruation.4 The theory of retrograde cell trafficking has also been considered a major etiology that may explain why ovarian endometriosis is the precursor of 20 % of ovarian cancers.27 In the absence of visible endometriosis, the removal of the tubes and ovaries is an elective decision made based on patient history. There is literature that strongly suggests that hysterectomy alone may not treat the patient’s symptoms of pain due to unrecognized disease left behind (incomplete surgery).23
Some researchers believe that endometriosis can be present from birth – a theory separate from retrograde menstruation. The embryological concept is more probable because cells, which are supposed to end up inside the uterus, sometimes stay in ectopic locations.5,26 Excision can definitively cure such cases of rectovaginal endometriosis of embryologic origin, in contrast with endometriosis due to implantation of retrograde menstrual blood, which may continue to implant. Cases of rectovaginal endometriosis are considered to be the ‘most painful’ and are often diagnosed just from rectovaginal examination. In 1914, Thomas Cullen said, ‘the removal of extensive adenomyoma of the rectovaginal septum (deep endometriosis surgery) is infinitely more difficult than a hysterectomy (Wertheim) for carcinoma of cervix procedure‘.9 Today, most gynecologic oncology surgeons agree.
4.8.1.1 Is there a Cure for Endometriosis?
My colleague, Dr. Harry Reich, does not strongly believe in the implantation and retrograde menstruation theory. He has said that, ‘If endometriosis came back every month, there would be no point in doing surgery. The endometriosis cells in many women have been around since birth. These cells begin a cycle of chronic inflammation on a monthly basis starting with menstrual periods. Fibromuscular tissue is deposited around the endometriosis glands and connective tissue with resulting pain. If diagnosed early, this tissue can be excised completely using the laparoscope. Unfortunately this type of surgery sometimes may include bowel resection and resection of small portions of upper vagina behind the cervix. Most of these cases can be diagnosed by a simple rectovaginal exam in the doctor’s office. MRI is rarely indicated.‘
Many patients’ fears are based on the idea that the disease will worsen and recur, thus becoming a chronic disease. They believe that the risk of recurrence is very high. In fact, no clinical data exists to determine whether endometriosis is a progressive and/or a recurrent disease, and many recurrent clinical symptoms may be explained by inadequate response to medical treatments and/or incomplete surgical excision of the disease.
Dr. Reich believes that true recurrence of the disease is rare. In most cases, the surgical excision patient’s symptoms do not get much worse after the initial surgical excision diagnosis. ‘Clinical recurrences’ can often be explained by incomplete or inappropriate treatments. Most ‘recurrences’ occur in areas involved during the initial surgical procedure. A study of second-look laparoscopy showed that recurrent disease occurs most frequently at the site of treated disease after surgical treatment of early endometriosis, reflecting incomplete excision or ablation of disease.7 After treatment of colorectal endometriosis, most re-operated patients are treated in the same area, suggesting that part of the disease was left behind due to an inadequate surgical procedure or because some organs were undertreated to minimize the risk of postoperative complications.
In addition, intentionally leaving endometriosis tissue behind and expecting that, by simply removing the ovaries, the lesions will begin an inactive phase, is a misappropriation of decisionmaking. In the presence of diffuse fibrosis of the peritoneum and unrecognized deep nodular and infiltrative disease, definitive surgery limited to simple total hysterectomy and bilateral salpingo-oophorectomy (BSO) is insufficient.
4.8.1.2 Indications for Hysterectomy
The surgical choice for treatment of endometriosis depends on many factors, including the patient’s age, severity of her symptoms, and whether she wants to have children. These are all key factors in decision-making. A thorough history and physical exam is warranted for determining many of these factors, as well as gauging the areas that may be affected in the pelvis. A hysterectomy alone will not definitively cure endometriosis if the tissues with endometriosis in the pelvis are not also removed. Removal of the uterus should have justifiable indications. Pelvic pain, after all, may have multiple causes. Therefore, in the absence of uterine disease, doctors and patients should be cautious in proceeding with hysterectomy. In addition, preoperative tests like MRIs do have false sensitivity in detecting uterine pathology such as endometriosis or adenomyosis.3
4.8.2 Uterine Pathology
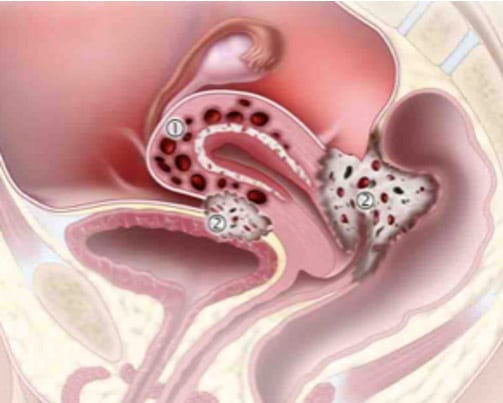
The most common uterine pathologies seen with endometriosis are adenomyosis, leiomyomata uteri, adhesions, and deep fibrotic disease involving posterior cul-de-sac, as well as anterior cul-de-sac involving the bladder. All of the above pathologies, particularly adenomyosis, are characterized by severe dysmenorrhea, menorrhagia, and pelvic pain. Adenomyosis is the thickening of the endo-myometrial junction and the extension of glands deep into the myometrium from the endometrial cavity (Fig. 4.31). However, the uterus can also be infiltrated by the presence of disease in neighboring organs through the serosal layer.
In advanced endometriosis, the presence of an ovarian endometrioma, a harbinger of retroperitoneal disease and deep nodular and infiltrative endometriosis, is one of the most important factors in determining severity and extent of pathology. An intraperitoneal spillage of chocolate cyst contents from an endometrioma may cause severe adhesions to the ovaries, the uterus, the pelvic sidewalls, the rectosigmoid, and the cul-de-sac. An endometrioma spillage may also occur retroperitoneally, causing severe fibrosis involving the ureter and the nerves.
4.8.3 Preoperative Considerations
The disease can involve multiple pelvic organs, mainly the uterus, ovaries, tubes, bowel, and urinary tract, along with retroperitoneal spaces. The extra-uterine involvement determines the outcome of the planned procedure. Many of these patients, having had previous laparoscopic procedures, can present a wide range of symptoms suggestive of multiple organ involvement. Patients must be aware that endometriosis surgery carries 5–10 times more complications and risks than simple laparoscopic procedures.8 In order to obtain a proper consent, the best practice is to obtain a preoperative consultation with all the specialists in the surgical team. Outcome correlates positively with the experience and the skill of the surgeon and his or her team whose focus is exclusively endometriosis surgery.24
When extensive cul-de-sac involvement with endometriosis is suspected, either clinically or from another doctor’s operative record, a mechanical bowel preparation should be considered prior to surgery. Polyethylene glycol-based isoosmotic solution (GoLYTELY or Colyte) works well. Following dissolution in water to a volume of 4 liters, oral administration induces a diarrhea that rapidly cleanses the bowel (generally within 4 hours). GoLYTELY is usually taken the afternoon before surgery so as not to interfere with sleep. Reglan (metoclopramide), 10 mg, is administered 30 minutes before the bowel prep to help promote gastric emptying and thus reduce abdominal bloating and distension.
4.8.3.1 Hysterectomy Technique
The treatment of endometriosis is the excision of all endometriotic lesions. For definitive treatment of endometriosis when hysterectomy is indicated, the operation should be tailored to relieve all of the symptoms, not only the symptoms of suspected uterine origin. All extrauterine superficial and deep endometriosis should be excised first before the hysterectomy. The operation typically begins with adhesiolysis, followed by excision of endometriosis, after which hysterectomy is the final stage.
Hysterectomy is performed using an extra-fascial technique as opposed to an infra-fascial technique. In the extra-fascial approach, retroperitoneal exposure with the identification of ureter and the uterine vessels is essential. Similarly, parametrial ligaments, pericervical rings, and vaginal fornices are approached by using the extra-fascial technique to mobilize the bladder and rectum.
4.8.3.2 Extrafascial Hysterectomy
An extra-fascial approach to hysterectomy for endometriosis often involves similar techniques used for modified radical hysterectomy for carcinoma of cervix. This extended version of modified hysterectomy is also referenced as a tailored radical hysterectomy for endometriosis.12 In multiple organ involvement due to deeply infiltrating endometriosis (DIE), hysterectomy may be even more complex and technically more difficult than radical hysterectomy. Because of DIE, there is usually a more frequent necessity to repair the bowel, bladder, and ureter. It is key to have a very thorough understanding of pelvic retroperitoneal anatomy and an experienced strategy for hemostasis in identifying the uterine arteries from their origin before dividing the uterine ligaments and vaginal attachments.
At the start of surgery, regardless of the extent of the pelvic disease, one always begins with thorough inspection of the upper peritoneal cavity, the liver, and both diaphragmatic reflections, as well as careful inspection of the small bowel, the appendix, and colon. Steep Trendelenburg positioning of the patient with the uterine manipulator in place is very important for the exposure and visualization of the pelvis. Introduction of a rectal probe at this initial stage may be helpful and failure to advance it may be a sign of narrowing.
Before hysterectomy is attempted, all issues regarding a safe procedure are planned and methodically exercised. After the patient’s pelvic pathology is fully evaluated, the plan of surgery will be to mobilize and take the steps forward cautiously and patiently, from the easier to the more difficult steps. Many of the retroperitoneal spaces may be obliterated with infiltration by deep fibrotic disease and may not be identifiable or recognized. Due to endometriosis, these spaces anatomically are lost to retroperitoneal fibrosis.
4.8.3.3 Pelvic Adhesiolysis
The first step is to free the uterus encased by surrounding adhesions and fibrotic endometriosis, along with freeing all bowel loops in the pelvis. Any small bowel that is attached to the vesicouterine peritoneal fold, uterus or the rectum should be liberated. There are three key points when performing intestinal adhesiolysis within the pelvis:
- Scissors dissection without electrosurgery;
- Countertraction;
- Blunt dissection.
The bowel is gently held with an atraumatic grasper and lifted away from the structure to which it is adhered, exposing the plane of dissection. When adhesive interfaces are obvious, scissors are used. The blunt-tipped scissors are used to sharply dissect the adhesions in small, successive cuts taking care not to damage the bowel serosa. Countertraction will further expose the plane of dissection and ultimately free the attachment. Electrosurgery and laser are generally not used for adhesiolysis involving the bowel due to the risk of thermal damage and recurrent adhesion formation. However, when adhesive aggregates blend into each other, initial incision is made very superficially with laser, and aquadissection distends the layers of the adhesions, facilitating identification of the involved structures. Division of adhesions continues with laser. The aquadissector and injected fluid from it are used as a backstop behind adhesive bands that are divided with the CO2 laser.17
The rectosigmoid is often adherent to the left pelvic sidewall obscuring visualization of the left adnexa. Dissection starts well out of the pelvis in the left iliac fossa. Scissors are used to develop the space between the sigmoid colon and the psoas muscle to the iliac vessels, and the rectosigmoid is reflected toward the midline. Thereafter, with the rectosigmoid placed on traction, rectosigmoid and rectal adhesions to the left pelvic sidewall are divided starting cephalad and continuing caudad.
The round ligaments are bisected, and the incision is carried toward the midline vesicouterine fold. The vesicouterine fold is a common location of deep fibrotic endometriosis extending to full thickness into the bladder and the uterine wall. In a limited number of cases, when the bladder is completely frozen with adenomyosis, the need for bladder resection will be part of the extended hysterectomy procedure. Therefore, paired paravesical spaces will be further extended anteriorly into the Retzius space to mobilize the bladder anteriorly.
4.8.3.4 Ureterolysis
Mobilization of rectosigmoid colon helps to gain access to the left retroperitoneum and in identifying the ureters at the pelvic brim. Using atraumatic forceps, the infundibulopelvic (IP) ligament is lifted anterolaterally and the ureter is identified medially at the pelvic brim as it crosses the common iliac artery.
The ureters will also be recognized after division of the round ligaments, and extending the incision which is carried cranially parallel and lateral to the ovarian vessels and adnexa on the roof of broad ligament. With traction on the adnexa medially, the external iliac major vessels will be exposed laterally. The ureter again will be at the medial leaf of the peritoneum above the bifurcation of the common iliac artery and 1 to 3 cm below the IP ligament. Particular attention should be paid advancing into the paired paravesical and paired pararectal spaces as it is essential in isolation of both uterine arteries and the ureters.
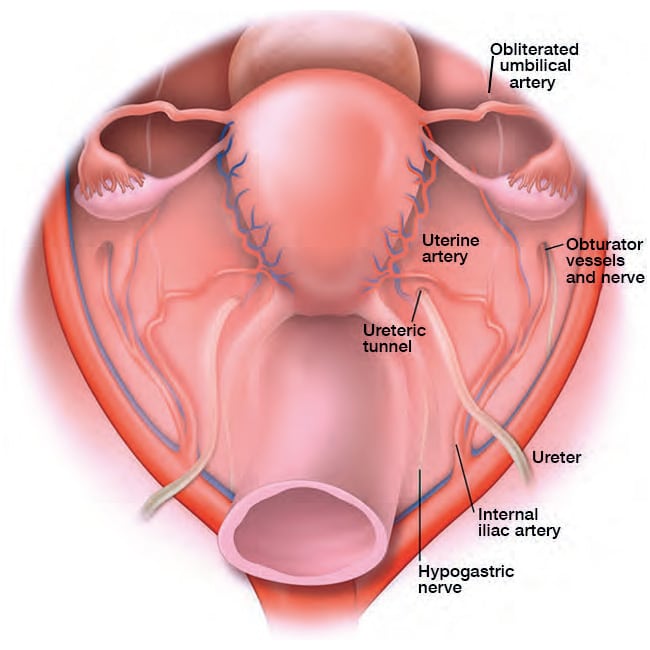
The critical objective at this step is to trace the ureters caudally to the level of the uterine artery bridge, also known as ureteric tunnel (Fig. 4.32). During the course of the dissection of the ureter from pelvic brim, the surgeon recognizes the internal iliac artery branching into the hypogastric division and obliterated umbilical artery lateral to the course of the ureter (Fig. 4.32). When deep nodular fibrosis involves the retroperitoneum at the ovarian fossa, the peritoneum making up the pelvic sidewall facing the ovarian endometrioma is thickened, contracted and deformed with inflammatory fibrosis. Using the fine-tipped Adson forceps (Reich modification), diseased peritoneum is lifted upwards and medially retracted and then excised to unroof the ovarian fossa medially to the uterosacral plate. Fibrotic endometriosis may involve deeper levels beyond the adventitia, as the ureter may well be encased completely with overlying uterine artery in the ureteric tunnel.
Due to unpredictable nature of the extent of fibrosis, ureteral stents may help reduce surgical time and prevent complications at this step. The difficulty of identifying the ureters by tracing them to the ureteric tunnel depends on the extent of the fibrosis. After the excision of the disease, the ureter can be repaired primarily, or end-to-end anastomosed with fine suturing. Hydrodistention of the peritoneum using contrast with methylene blue may aid in both identification of endometriotic lesions that otherwise may be left out and as well as increasing the safety of the procedure.
4.8.3.5 Hypogastric and Uterine Artery Ligation
In order to avoid hemorrhage, the hypogastric and uterine artery can be ligated anywhere from its origin towards its course to the uterus. The uterine artery is identified by tracing the common iliac to internal iliac artery, and to hypogastric artery distally or by locating the obliterated umbilical artery and tracing it proximally.10
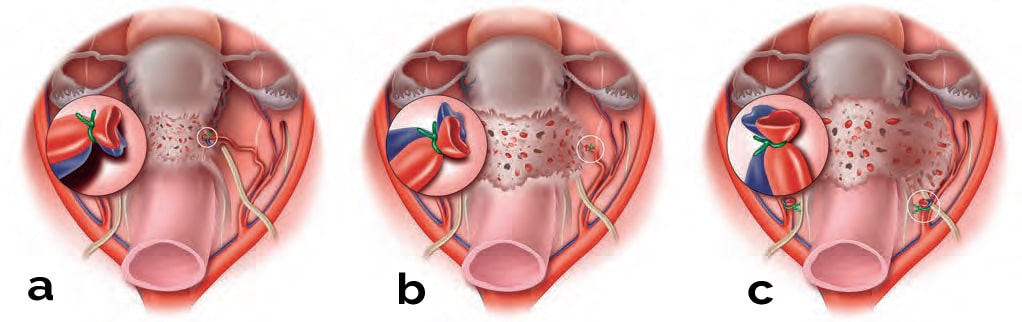
Depending on the involvement of the parametrial tunnel and the vesico-uterine pillars with fibrotic nodular endometriosis, the uterine arteries are ligated at any point from its origin from the common iliac artery during its course towards the uterus. Three levels of uterine artery ligation are described here. Level one is the ligature of the uterine artery at the segment from the ureteric tunnel to the uterine junction (Fig. 4.33a). Level two is ligation between the obliterated umbilical ligament and the ureteric tunnel (Fig. 4.33b). Finally, if disease is extending to the obturators and to the hypogastric reflection, level three ligation at the level of the internal illiac artery may be performed (Fig. 4.33c). Another alternative is to implement suture ligation using the anterior approach from the prevesical space in cases where the posterior compartment is frozen.
When the ovarian vessels are ligated bilaterally and there are well-placed sutures on the uterine arteries, the uterus should blanch and change color immediately. Suture ligation is always preferred over energy-based modalities, like the harmonic scapel and bipolar electrocautery. The procedure will then proceed by dissecting the posterior cul-de-sac.
4.8.3.6 Peritoneal Implants; Focal Excision and Peritonectomy
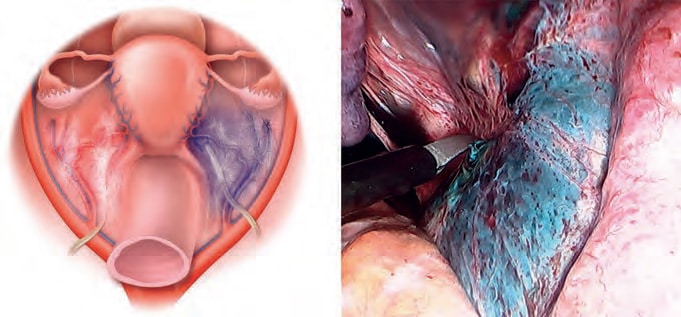
In order to treat superficial peritoneal endometriosis, endometriotic implants and their adjacent peritoneum can be excised using scissors or a needle electrode with a cutting current. An elliptical incision is made around the lesion, its edge lifted upward, and the lesion is undermined using the hydraulic effect of pressurized irrigant from an laparoscopic suction-irrigation pump (HAMOU Endomat®, KARL STORZ Tuttlingen, Germany). This pushes pelvic sidewall structures and the rectum away and allows for safer and easier undercutting of the lesion. Following excision with scissors, the ureter, the anterior rectal wall, and the upper posterior vagina are checked and superficial endometriosis in these areas are excised. A blue dye contrast may aid the surgeon to detect occult and atypical peritoneal lesions (Aqua Blue Contact Technique) (Figs. 4.34).25
Small pinpoint lesions can be excised using scissors, the CO2 laser or electrosurgery with the resultant drainage of haemosiderin-filled fluid in cases where deposits have infiltrated beneath the peritoneum. The base of the lesion must also be excised until normal tissue is seen. Cold scissors with microbipolar back-up may result in better outcome due to their greater depth of access and ability to restore normal anatomy.
In cases of endometrioma, the anti ovarian peritoneal surface is largely involved in the disease. Peritoneum is thickened and contracted due to the fibrosis that extends deeper into pelvic sidewall and cul-de-sac. To treat deeper and wider peritoneal endometriosis, after the ovary is freed, a peritonectomy is performed where the peritoneum of the pelvic sidewall is excised to the extent of involvement with endometriosis. Using methylene blue water to enhance color contrast in the hydrodistention underneath the peritoneal layer may assist the surgeon in recognizing deeper implants. With blunt and sharp dissections, the uterine artery is skeletonized as the diseased tissue is retracted to the midline. After the identification of the uterine artery, the diseased peritoneum is excised all the way to the uterine artery tunnel.
At this juncture, the uterosacral ligament and its presacral attachments, which is the reflection of the inferior hypogastric nerves, is deferred and the pararectal space is identified in order to expose the extent of rectovaginal involvement.
When the rectal probe advances into the rectum and further into the rectosigmoid, countertraction is created. At the same time, the uterus is anteverted with its manipulator. The surgeon uses the blunt scissors to separate the rectum from its posterior uterine and posterior cervical and vaginal involvement.
4.8.4 Endometriomas
A suction-irrigation pump (HAMOU Endomat®, KARL STORZ Tuttlingen, Germany) is used to lift up the ovaries if they are attached by adhesions to their respective utero-sacral ligament and/or pelvic sidewall. Often, this maneuver will result in drainage of an endometrioma from the undersurface of the ovary. If no endometrioma is readily identified, and the patient has ‘unexplained infertility’ or pre- or post-menstrual spotting, a 3-mm knife electrode connected to unmodulated unipolar cutting current (80 W) is used to incise and drain areas on the ovary with superficial endometriosis and cysts suspicious for endometrioma. If an endometrioma is discovered by either of these two methods, the cyst cavity is rinsed with Ringer’s lactate solution and then excised using 3-mm and 5-mm biopsy forceps, grasping forceps, and/or scissors. To help delineate the initial plane between normal ovarian cortex and endometrioma cyst wall, cutting current through the knife electrode tip applied at the cyst wall-cortex junction results in the development of a dissection plane. This step is particularly useful near the utero-ovarian ligament as rough avulsion can lead to excessive bleeding. Grasping forceps are then used to stabilize ovarian cortex while the endometrioma cyst wall is avulsed. Excision can be performed with minimal bleeding from the cyst wall bed and the ovarian wall edges usually reapproximate quite well, though occasionally extracorporeal suturing is required.19 Finally the pelvic sidewall peritoneum where the ovary was attached usually overlying the ureter is excised.
4.8.4.1 Partial and Complete Cul-de-sac Obliteration
- Cul-de-Sac Excision
Cul-de-sac obliteration implies the presence of deep fibrotic
endometriosis.
Partial cul-de-sac obliteration means that deep fibrotic endometriosis, i.e., endometriosis beneath the peritoneum, is present and is severe enough to alter the course of the rectum. The deep fibrotic endometriosis is usually located on the rectum, in the rectovaginal space, on the upper vagina, in the space between the upper vagina and the cervix (cervicovaginal angle), or in one or both utero-sacral ligaments. With deep culde- sac obliteration, fibrotic endometriosis and/or adhesions involve the entire area between the cervico-vaginal junction (and sometimes above) and the recto-vaginal septum. Often one area predominates.20
Careful inspection of the cul-de-sac is necessary to evaluate the extent of upward tenting of the rectum. To determine if culde- sac obliteration is partial or complete, a sponge on a ring forceps is inserted into the posterior vaginal fornix. Complete cul-de-sac obliteration implies that the outline of the posterior fornix cannot be visualized initially through the laparoscope, i.e., the rectum or fibrotic endometriosis nodules completely obscure the identification of the deep cul-de-sac. Partial culde- sac obliteration occurs where rectal tenting is visible but a protrusion from the sponge in the posterior vaginal fornix is noted between the rectum and the inverted ‘U’ of the uterosacral ligaments.
In contrast to the procedure performed for superficial peritoneal endometriosis, deep fibrotic nodular endometriosis involving the cul-de-sac, often with invasion into posterior vagina, rectum, or posterior cervix, is a much more difficult problem and should be attempted only by the most expert laparoscopist. At this step the ureter is completely exposed bilaterally. First, the posterior uterus will be freed from its rectosigmoid. Then, following the placement of rectal, vaginal, and uterine probes, the rectal serosa is opened at its junction with the cul-de-sac lesion using scissors. Rather than concentrating on the excision of the nodular mass, attention is first directed to the complete dissection of the anterior rectum throughout its area of involvement. The cul-de-sac is approached with sharp dissection by scissors, avoiding any usage of energy-based instruments except the bipolar for hemostasis. Dense adhesions and fibrotic nodules are freed from the uterine border, leaving most of the disease above the rectum on the posterior vagina, especially if the surgeon can visualize the outline of the rectal probe during the dissection. The dissection is performed until the loose areolar tissue of the recto-vaginal space is reached. Usually, this dissection extends 3–4 centimeters below the posterior fornix. Another technique in reaching the rectovaginal disease is performed by exploring the pararectal space bilaterally. Only after the rectum is mobilized should excision of the fibrotic endometriosis be attempted from the rectum, posterior vagina, and utero-sacral ligaments.
When a ureter is close to the lesion, its course in the deep pelvis is traced by opening its overlying peritoneum with scissors. On the left, this often requires scissors reflection of the rectosigmoid, as previously described, starting at the pelvic brim. Microbipolar forceps are used to control arterial and venous bleeding.
The rectum, with or without a fibrotic lesion must be separated from the posterior uterus and upper vagina, especially when operating close to the uterine vessels, as entry into these vessels can produce life-threatening haemorrhage requiring immediate application of the bipolar forceps.
Following separation of the rectum from the back of uterus and the upper posterior vagina, the dissection continues on top of the posterior vagina, the position of which is confirmed by the sponge in the posterior fornix. This dissection on the outside of the vaginal wall uses laser, aquadissection, electrosurgery or scissors. On occasion, the lesion may extend deep into or completely through the vaginal wall. Dissection should be performed accordingly with removal of all visible fibrotic endometriosis. Lesions extending totally through the vagina demand an en bloc resection from cul-de-sac to posterior vaginal wall as part of the extended hysterectomy. At this stage, the surgical team may also perform a low anterior rectal resection if rectal stricture is present, or the anterior surface of the rectum is compromised due to wide excision.
Our recommendation is to defer the low anterior segmental resection until after the hysterectomy is completed. The reason for this is that the natural orifice of the vagina could be used as an access route for the resected bowel to be removed.13
4.8.4.2 Rectum
Endometriosis nodules infiltrating the rectal muscularis are excised, partially or totally, usually with the operator’s or the assistant’s finger, in the rectum just beneath the lesion. Some endometriosis nodules in the muscularis of the anterior rectum can be excised laparoscopically without entering the rectum. Full-thickness penetration of the rectum can occur during hysterectomy surgery, especially when excising rectal endometriosis nodules. Deep rectal muscularis defects should always be closed with suture. These defects are detected by filling the rectum and rectosigmoid with a blue dye solution. Full thickness rectal lesion excisions can be repaired by laparoscopic suture or incorporated in an anterior discoid resection using a # 29 or 33 EEA stapler. Following identification of the nodule or rent in the rectum, a closed circular stapler [Proximate ILS Curved Intraluminal Stapler (Ethicon, Stealth)] is inserted into the lumen just past the lesion or hole, opened 1–2 cm, and held high to avoid the posterior rectal wall.21 The proximal anvil is positioned just beyond the lesion or hole, which is invaginated into the opening, and the device closed. The instrument is fired and removed. If bowel resection is deemed necessary, it will be performed before the vaginal cuff is closed. In order to prevent complications, surgeons are cautioned against the use of active suction drains or mechanical suction drain pumps. The walls of pelvic organs which are already compromised and thinned due to extensive shaving and excision may be prone to leakage of the anastomosis or fistula formation. In addition, juxtapositional alignment of suture repair lines must be avoided when full thickness organs, such as the ureter, bowel, bladder and vagina, are simultaneously repaired. Mobilizing an omental pedicle pad in between the repaired organs may also be considered in order to prevent rectovaginal, uretrovaginal, vesicorectal, and vesicovaginal fistula formation.
When very low anterior bowel reanastomosis is performed or a compromised bowel wall tissue is suspected, a prophylactic temporary loop ileostomy is advised.1 In cases when ureter re-anastomosis or re-implantation is performed, prolonged use of post-operative ureteral stents are necessary.
4.8.4.3 Parametrium and Frozen Pelvis
When endometriosis involves the parametrium, the uterine and bowel may also be involved. This can result in a partially frozen pelvis. In this surgically challenging situation, the uterine arteries are ligated proximally at the hypogastric level or the internal iliac artery level. Stricture of the ureter and bowel is not uncommon when the uterus is partially frozen to the pelvic sidewall. The detachment of deep fibrotic tissue from parametrial extension in frozen pelvis is the most challenging aspect of a modified radical hysterectomy. These lesions may extend to the level of the obturator vessels and deep into ischial spine.
It is again very important to have pre-operatively placed ureteral stents since the ureters are almost always encased within this fibrotic process. Any muscular defect involving the ureter during this excision should be immediately repaired with 4.0 Proline or Monocryl sutures in an interrupted fashion, whereas primary lesions on the rectum are closed with interrupted silk in a single layer. After the disease-free tissues are reached, both anteriorly and posteriorly, the cardinal ligaments are taken down either with a harmonic scalpel or pure cut electrical current.
4.8.4.4 Uterosacral Ligaments
Removal of uterosacral ligaments with its presacral attachment and deep hypogastric extensions is again performed since the disease usually affects the proximal part of the uterosacral ligaments. However, deep nodules and cystic lesions may involve the pararectal space extending into the presacral and hypogastric compartments. Extreme caution must be practiced as well as avoidance of bilateral deep excisions in order to prevent the potential for serious bladder dysfunction secondary to injury of the deep hypogastric nerve.
- Division of Cervicovaginal Attachments and Circumferential
Culdotomy
The cardinal ligaments and uterosacral ligaments on each side are divided lateral to endometriotic fibrosis. Bipolar forceps coagulate the uterosacral ligaments. The vagina is entered posteriorly over the uterovaginal manipulator below the cervicovaginal junction. A 4-cm-diameter reusable vaginal delineator tube is placed in the vagina to prevent loss of pneumoperitoneum and to outline the cervicovaginal junction circumferentially as it is incised using the CO2 laser, harmonic scalpel, or electrosurgery to complete the circumferential culdotomy with the delineator as a backstop. The specimen of the uterus with its contiguous tissues (posterior vagina and parametrium, along with tubes and ovaries attached) is removed from the vagina. The uterus is morcellated vaginally, if necessary, and pulled out of the vagina.
Extrafascial culdotomy with removal of the entire uterus is followed with anchoring of the vaginal cuff to the uterosacral ligaments.
Intrafascial culdotomy proximal to the uterosacral ligament insertion site preserving Level 1 support will promote future pelvic organ prolapse surgery and should never be done if endometriosis exists in the cul-de-sac. A supracervical hysterectomy will do the same! Culdotomy proximal to the uterosacral ligament insertion site preserving Level 1 support is more like a supracervical hysterectomy than a TLH. More important, the uterosacral insertions are the most common site of deep endometriosis, and, if left behind, may contribute to future pelvic pain and dyspareunia problems.
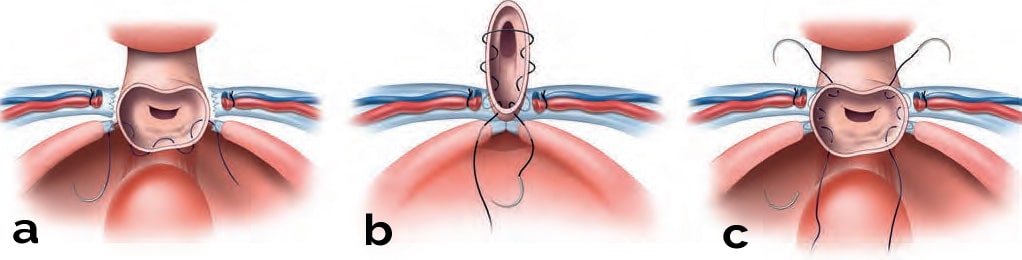
- Laparoscopic Vaginal Vault Closure and Suspension with McCall
Culdoplasty
The vaginal delineator tube is placed back into the vagina for closure of the vaginal cuff, occluding it to maintain pneumoperitoneum, and hemostasis around the vaginal cuff obtained using microbipolar forceps with irrigation. The uterosacral ligaments are identified by bipolar desiccation markings or with the aid of a rectal probe. The first suture is complicated as it brings the uterosacral and cardinal ligaments, as well as the rectovaginal fascia, together. This single suture is tied extracorporeally, bringing the uterosacral ligaments, cardinal ligaments, and posterior vaginal fascia together across the midline. It provides excellent support to the vaginal cuff apex, elevating it and its endopelvic fascia superiorly and posteriorly toward the hollow of the sacrum. The rest of the vagina and overlying pubocervicovesicular fascia are closed vertically with one or two 0-Vicryl interrupted sutures. Cuff closure sutures are for fascia and not vaginal epithelium. They are used for support, not hemostasis. Strangulating sutures that inhibit tissue circulation are avoided. A drain is never used. Locked sutures are a cause of both tissue strangulation and poor drainage and are never used.
The cuff may be closed vertically or horizontally depending on whether the patient needs the support of the posterior vagina. The modified McCall culdoplasty procedure is performed when the cuff is closed vertically in two layers (Figs. 4.35a, b). When the cuff is closed horizontally, modified Richardson angle sutures are applied (Fig. 4.35c). In this technique, interrupted angle sutures are applied.
4.8.5 Cystoscopy
Cystoscopy is done after vaginal closure to check for ureteral patency in most cases, after intravenous administration of indigo carmine dye. This is necessary when the ureter is identified but not dissected and especially necessary when the ureter has not been identified. Blue dye should be visualized through both ureteral orifices. The bladder wall should also be inspected for suture and thermal defects.22
4.8.5.1 Underwater Examination
At the close of each operation, an underwater examination is used to detect bleeding from vessels and viscera tamponaded during the procedure by the increased intraperitoneal pressure of the CO2 pneumoperitoneum. The CO2 pneumoperitoneum is displaced with 2–4 L of Ringer’s lactate solution, and the peritoneal cavity is vigorously irrigated and suctioned until the effluent is clear of blood products. Any further bleeding is controlled underwater using microbipolar forceps to coagulate through the electrolyte solution, and 2 L of lactated Ringer’s solution is left in the peritoneal cavity.
4.8.5.2 Skin Closure
The vertical intra-umbilical incision is closed with a single 4-0 Vicryl suture opposing deep fascia and skin dermis, with the knot buried beneath the fascia. This will prevent the suture from acting like a wick transmitting bacteria into the soft tissue or peritoneal cavity. The lower quadrant 5-mm incisions are loosely approximated with a Javid vascular clamp (V. Mueller, McGaw Park, Illinois, USA) and covered with Collodion (AMEND, Irvington, New Jersey, USA) to allow drainage of excess Ringer’s lactate solution.
4.8.6 Summary
Extensive stage IV endometriosis can be excised laparoscopically if the surgeon is willing to spend the time to do so. Hysterectomy usually results in relief of the patient’s pain, including symptoms related to enlarged uterus and menorrhagia. Oophorectomy may not be necessary during hysterectomy for advanced endometriosis if the endometriosis is removed carefully.16
When hysterectomy is considered as the permanent solution for endometriosis patients, complete removal of the diseased tissue and the cervix is only feasible by implementing the extrafascial technique. A skilled and tenacious surgical team, consisting of colorectal and urology specialists and led by a gynecological surgeon who exclusively deals with endometriosis, is of paramount importance for the best longterm treatment of endometriosis.
References
- Akladios C, Messori P, Faller E, Puga M, Afors K,
Leroy J et al. Is ileostomy always necessary following rectal resection for deep infiltrating endometriosis? J Minim Invasive Gynecol 2015;22(1):103–9. doi:10.1016/j.jmig.2014.08.001. - American College of Obstetricians and Gynecologists . Salpingectomy for Ovarian Cancer Prevention: Committee Opinion No. 620. Obstet Gynecol 2015(125):279–81.
- Bazot M, Darai E, Hourani R, Thomassin I, Cortez A, Uzan S et al. Deep pelvic endometriosis: MR imaging for diagnosis and prediction of extension of disease. Radiology 2004;232(2): 379–89. doi:10.1148/radiol.2322030762.
- Benagiano G, Brosens I, Habiba M. Structural and molecular features of the endomyometrium in endometriosis and adenomyosis. Hum Reprod Update 2014;20(3):386–402. doi:10.1093/humupd/dmt052.
- Bou quet dJJ, Ayoubi JM, Lesec G, Validire P, Goguin A, Gianaroli L et al. Identification of displaced endometrial glands and embryonic duct remnants in female fetal reproductive tract: possible pathogenetic role in endometriotic and pelvic neoplastic processes. Front Physiol 2012;3:444. doi:10.3389/fphys.2012.00444.
- Brosens I, Benagiano G. Endometriosis, a modern syndrome. Indian J Med Res 2011;133:581–93.
- Canis M, Bourdel N, Houlle C, Gremeau AS, Botchorishvili R, Matsuzaki S. Endometriosis may not be a chronic disease: an alternative theory offering more optimistic prospects for our patients. Fertil Steril 2016;105(1):32–4. doi:10.1016/j.fertnstert.2015.09.009.
- Cicco C de , Corona R, Schonman R, Mailova K, Ussia A, Koninck x P. Bowel resection for deep endometriosis: a systematic review. BJOG 2011;118(3):285–91. doi:10.1111/j.1471-0528.2010.02744.x.
- Cullen TS. Adenomyoma of the rectovaginal septum. Journal of the American Medical Association 1914;LXII(11):835. doi:10.1001/jama.1914.02560360015006.
- Cundiff GW, Te Linde RW. Te Linde’s atlas of gynecologic surgery. Philadelphia, PA: Lippincott Williams & Wilkins; 2013. (ISBN No. 160831068X).
- Du H, Taylor HS. Contribution of bone marrow-derived stem cells to endometrium and endometriosis. Stem Cells 2007;25(8):2082–6. doi:10.1634/stemcells.2006-0828.
- Fedele L, Bianchi S, Zanconato G, Berlanda N, Borruto F, Frontino G. Tailoring radicality in demolitive surgery for deeply infiltrating endometriosis. Am J Obstet Gynecol 2005;193(1):114–7. doi:10.1016/j.ajog.2004.12.085.
- Fleisch MC, Hepp P, Kaleta T, Am Schulte EJ, Rein D, Fehm T et al. Feasibility and first long-term results after laparoscopic rectal segment resection and vaginal specimen retrieval for deep infiltrating endometriosis. Arch Gynecol Obstet 2014;289(6):1241–7. doi:10.1007/s00404-014-3146-3.
- Gargett CE, Schwab KE, Zillwood RM, Nguyen HP, Wu D. Isolation and culture of epithelial progenitors and mesenchymal stem cells from human endometrium. Biol Reprod 2009;80(6): 1136–45. doi:10.1095/biolreprod.108.075226.
- Gonzales M, Matos LA de , da Costa Gon çalves , Manoel Orlando , Blasbalg R, Dias Junior JA, Podgaec S et al. Patients with adenomyosis are more likely to have deep endometriosis. Gynecol Surg 2012;9(3):259–64. doi:10.1007/s10397-012-0746-4.
- Martin DC. Hysterectomy for treatment of pain associated with endometriosis. J Minim Invasive Gynecol 2006;13(6):566–72. doi:10.1016/j.jmig.2006.06.022.
- Reich H. Laparoscopic treatment of extensive pelvic adhesions, including hydrosalpinx. J Reprod Med 1987;32(10):736–42.
- Reich H. Issues surrounding surgical menopause. Indications and procedures. J Reprod Med 2001;46(3 Suppl):297–306.
- Reich H, McGlynn F. Treatment of ovarian endometriomas using laparoscopic surgical techniques. J Reprod Med 1986;31(7):577–84.
- Reich H, McGlynn F, Salvat J. Laparoscopic treatment of cul-de-sac obliteration secondary to retrocervical deep fibrotic endometriosis. J Reprod Med 1991;36(7):516–22.
- Reich H, Wood C, Whittaker M. Laparoscopic anterior resection of the rectum and hysterectomy in a patient with extensive pelvic endometriosis. Gynaecological Endoscopy 1998;7(2):79–83. doi:10.1046/j.1365-2508.1998.00162.x.
- Ribeiro S, Reich H, Rosenberg J, Guglielminetti E, Vidali A. The value of intra-operative cystoscopy at the time of laparoscopic hysterectomy. Hum Reprod 1999;14(7):1727–9.
- Rizk B, Fischer AS, Lotfy HA, Turki R, Zahed HA, Malik R et al. Recurrence of endometriosis after hysterectomy. Facts Views Vis Obgyn 2014;6(4):219–27.
- Ruffo G, Scopelliti F, Manzoni A, Sartori A, Rossini R, Ceccaroni M et al. Long-term outcome after laparoscopic bowel resections for deep infiltrating endometriosis: a single-center experience after 900 cases. Biomed Res Int 2014;2014:463058. doi:10.1155/2014/463058.
- Seckin TA, Newman NC, Seckin S. Histological Findings Following Excision of Peritoneal Endometriosis With and Without Using Aqua Blue Contrast Technique (ABCt™). J Minim Invasive Gynecol 2015;22(6):S52. doi:10.1016/j.jmig.2015.08.143.
- Signorile PG, Baldi F, Bussani R, D’Armiento M, Falco M de , Baldi A. Ectopic endometrium in human foetuses is a common event and sustains the theory of mullerianosis in the pathogenesis of endometriosis, a disease that predisposes to cancer. J Exp Clin Cancer Res 2009;28:49. doi:10.1186/1756-9966-28-49.
- Wei JJ, William J, Bulun S. Endometriosis and ovarian cancer: a review of clinical, pathologic, and molecular aspects. Int J Gynecol Pathol 2011;30(6):553–68. doi:10.1097/PGP.0b013e31821f4b85.
- Weir E, Mustard C, Cohen M, Kung R. Endometriosis: what is the risk of hospital admission, readmission, and major surgical intervention? J Minim Invasive Gynecol 2005;12(6):486–93. doi:10.1016/j.jmig.2005.09.001
Editors
- Tamer A. Seckin MD, FACOG, ACGE; Department of Obstetrics and Gynecology, Lenox Hill Hospital, New York NY, USA
- Serin Seckin MD; Department of Obstetrics and Gynecology, Mount Sinai St. Luke’s and Mount Sinai West, Icahn School of Medicine at Mount Sinai. New York NY, USA
- Harry Reich MD, FACOG, FACS; Wyoming Valley Health Care System, Wilkes- Barre, PA, USA; St. Vincent’s Medical Center,
Get a Second Opinion
Our endometriosis specialists are dedicated to providing patients with expert care. Whether you have been diagnosed or are looking to find a doctor, they are ready to help.Our office is located on 872 Fifth Avenue New York, NY 10065.
You may call us at (646) 960-3080 or have your case reviewed by clicking here.